abstract. Credit Transactions: Journal of the American Chemical Society (2024). DOI: 10.1021/jacs.4c03464
Trilayers are superior to bilayers in terms of fabrication and can improve the speed and capacity of electrochemical and electrocatalytic devices.
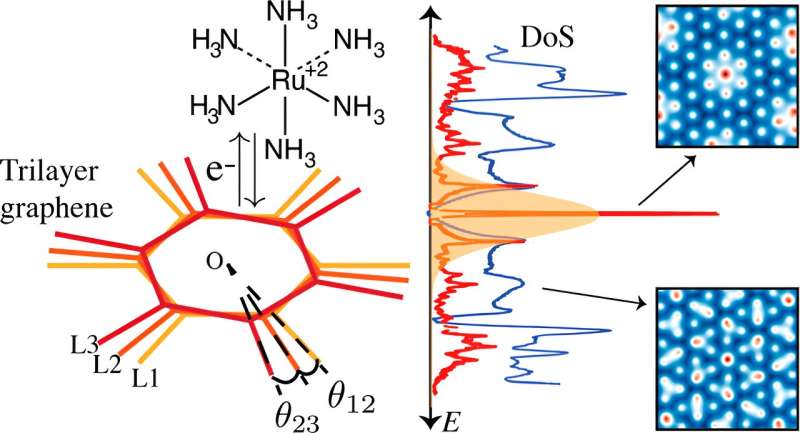
Three graphene layers in a twisted stack have the advantage of high conductivity, similar to “magic angle” bilayer graphene, but are easier to fabricate and allow faster electron transfer. The discovery could improve nanoelectrochemical devices or electrocatalysts to improve energy storage or conversion.
Graphene, a single layer of carbon atoms arranged in a hexagonal lattice, possesses unique properties including high surface area, excellent electrical conductivity, mechanical strength and flexibility, making this 2D material a strong candidate for increasing energy storage rate and capacity.
Twisting two sheets of graphene at a 1.1° angle, called the “magic angle,” creates a “flat band” structure. This means that across a range of momentum values, the electrons all have approximately the same energy. This results in a large peak in the density of states, or available energy levels that electrons can occupy, at the energy levels of the flat band, which improves electrical conductivity.
Recent research has exploited these flat bands to increase the charge transfer reactivity of twisted bilayer graphene when combined with an appropriate redox pair (a pair of chemicals often used in energy storage to move electrons between battery electrodes). It has been experimentally confirmed that this can be done.
A recent study by researchers at the University of Michigan found that adding additional graphene layers to create twisted triple-layer graphene enabled faster electron transfer compared to double-layer graphene, according to a model of electrochemical activity.
“We discovered highly flexible and enhanced charge transfer reactivity in twisted trilayer graphene that is not limited to specific torsion angles or redox pairs,” said Venkat Viswanathan, associate professor of aerospace engineering and corresponding author of the study. Journal of the American Chemical Society.
Stacking three layers of graphene creates an additional twist angle, creating a “non-normal” meaning that the pattern does not repeat at small angle twists, unlike double-layer graphene which forms a repeating pattern. Basically, if you add a third layer, the hexagonal grid won't align perfectly.
At room temperature, these non-repeating patterns have a wider range of angles with a higher density of states away from the flat band, resulting in an increase in electrical conductivity similar to that predicted from the magic angle.
“This discovery makes it easier to fabricate while avoiding the challenge of ensuring the exact twist angle required by bilayer graphene,” said Mohammad Babar, first author of the study and a PhD student in mechanical and aerospace engineering.
As a next step, the researchers plan to validate these findings experimentally and potentially discover even higher activities in multilayer twisted 2D materials for a wide range of electrochemical processes, such as redox reactions and electrocatalysis.
“Our work opens a new field of dynamics in 2D materials, capturing the electrochemical properties of suitable and not suitable structures. We can now identify the optimal balance of charge transfer reactivity in trilayer graphene for a given redox pair. said Babar.
Additional information:
Mohammad Babar et al., Twisto-electrochemical activity of trilayer graphene; Journal of the American Chemical Society (2024). DOI: 10.1021/jacs.4c03464
Provided by University of Michigan College of Engineering
Summons: Twisting and stacking three layers of graphene speeds up the electrochemical reaction (June 21, 2024). Retrieved June 22, 2024, from https://phys.org/news/2024-06-stacking-layers-graphene-electrochemical-reactions.html
This document is protected by copyright. No part may be reproduced without written permission except in fair dealing for personal study or research. The content is provided for informational purposes only.


