A team of researchers led by experts from the National Institute of Standards and Technology (NIST) has explained why the membranes that encapsulate cells have the ability to repel nano-sized molecules that approach them. Their findings could have implications for the development of several cell-targeted drug therapies.
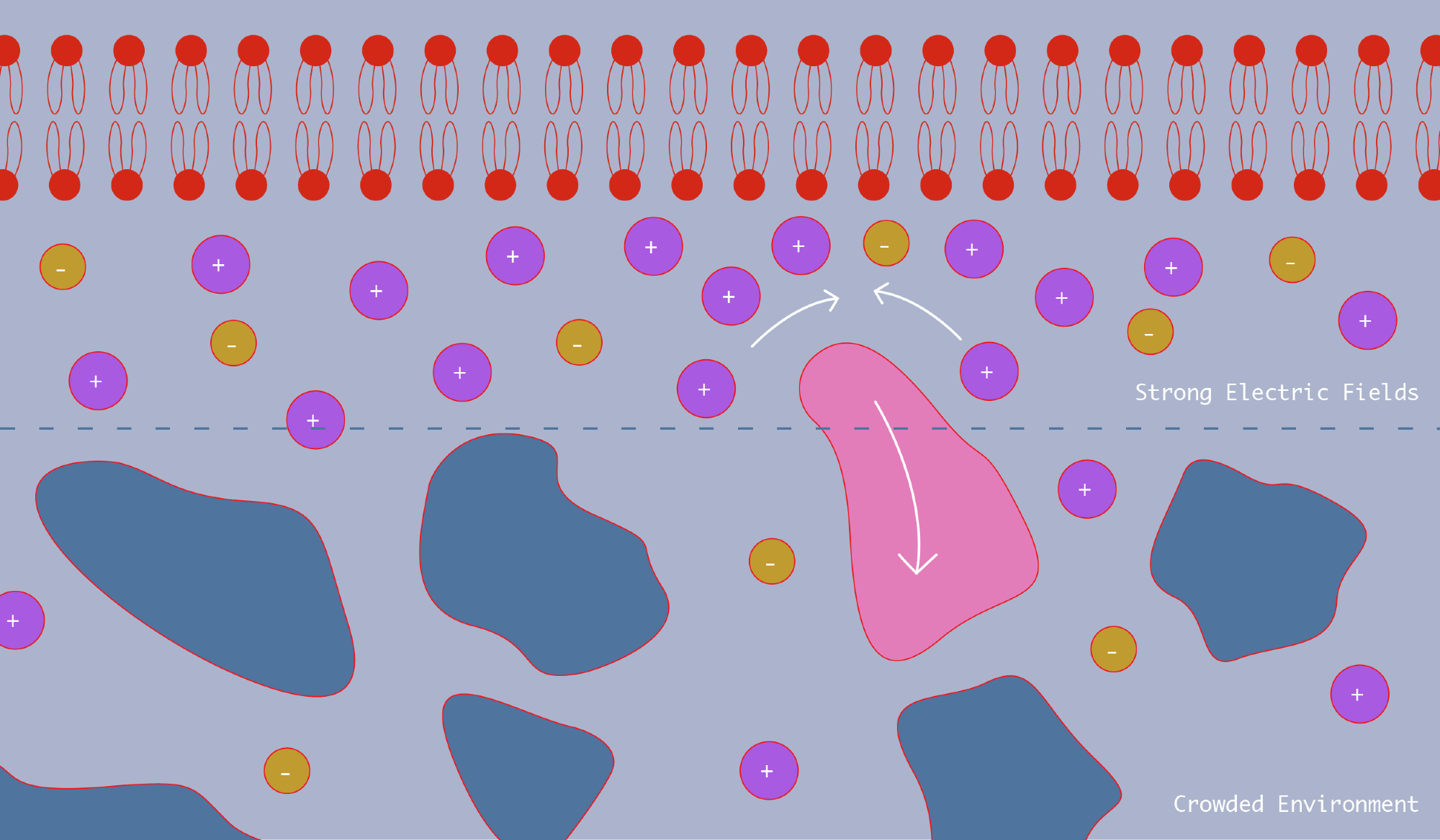
The team's results will be posted on: Journal of the American Chemical Society, demonstrate that the main mechanism preventing nano-sized particles from attaching to the cell surface is the strong electric field generated in the cell membrane. Neutral, uncharged nanoparticles are particularly affected by this repulsion force. In part, this is because the electric field attracts smaller charged molecules that disrupt the membrane and repel larger particles.
Repulsion may contribute to the efficacy of pharmaceutical treatments because many treatments are based on proteins and other nanosized particles that target membranes.
The results provide the first concrete evidence that the repulsive force is caused by an electric field. NIST's David Hoogerheide believes scientists should conduct further research into this effect.
These repulsive forces, along with the associated crowding exerted by smaller molecules, likely play an important role in how molecules with weak charges interact with biological membranes and other charged surfaces. This has implications for drug design and delivery and the behavior of particles in crowded environments at the nanometer scale..
David Hoogerheide, study author, NIST Neutron Research Center
Membranes define the boundaries of almost all cell types. In addition to the outer membrane that surrounds and protects the interior, cells have many membranes that make up components of organelles such as mitochondria and the Golgi apparatus.
Medical research benefits greatly from understanding membranes. One reason is that drugs often target proteins embedded in cell membranes. Certain membrane proteins act as gateways that control what enters and exits the cell.
There may be activity in areas close to this membrane. Cell membranes and thousands of different molecules are tightly packed together, and as anyone who has ever tried to break through that crowd knows, it can be difficult. Large molecules, such as proteins, have limited mobility, while small molecules, such as salts, can squeeze into tight spaces and move more easily.
Because molecular communities influence how cells work in the real world, this has become a very busy area of scientific research, Hoogerheide added. The careful interaction of the components in this cellular “soup” determines the function of the cell. It now appears that cell membranes may also be involved, as they sort nearby molecules based on their charge and size.
“How does crowding affect cells and their behavior? For example, how are the molecules in this soup sorted inside the cell so that some can be used for biological functions while others cannot? The effectiveness of the membrane can make a difference.”Hoogerheide added.
Despite the fact that scientists often use electric fields to move and separate molecules (a method known as dielectrophoresis), they have paid little attention to this effect at the nanoscale because moving nanoparticles requires very powerful fields. But the only thing an electrically charged membrane generates is a strong field.
Hoogerheide added:Electric fields near membranes in saline solutions, like our own, can be surprisingly strong. Its intensity drops off rapidly with distance, creating large field gradients that can repel nearby particles. So we used a neutron beam to investigate it.”
The scientists designed a test to investigate the effect of the membrane on nearby molecules of PEG, a polymer that produces chargeless nanoparticles, and how neutrons can distinguish between different isotopes of hydrogen.
Because hydrogen makes up a large part of PEG, the researchers were able to see how close the PEG particles were to the membrane by submerging the membrane and PEG in a solution of heavy water, which is created with deuterium rather than the hydrogen atoms normally found. water. They used equipment from Oak Ridge National Laboratory and NCNR's method known as neutron reflectometry.
Together with molecular dynamics simulations, this study provided the first evidence that significant field gradients in the membrane influence the repulsion force. That is, PEG molecules were much more repulsed from charged surfaces than from neutral surfaces.
Although the discovery does not reveal fundamentally new physics, Hoogerheide believes it demonstrates well-known physics in an unusual environment, which will pique the interest of scientists and prompt further investigation.
Hoogerheide concluded:We need to add this to our understanding of how things interact at the nanoscale. We have demonstrated the strength and importance of these interactions. Now we need to investigate what impact this has on the crowded environments where much of our biology takes place.”
Journal references:
Aguilella-Arjo, M.; From you. (2024) Charged biological membranes repel large neutral molecules by surface dielectrophoresis and counterion pressure. Journal of the American Chemical Society. doi:10.1021/jacs.3c12348
Source: https://www.nist.gov/


