Material synthesis and characterization
Firstly, we synthesized CoBiMn-LDH nanoparticles doped with different Bi content by a wet-chemical method. X-ray diffraction (XRD) patterns showed the characteristic (003) and (006) diffraction peaks of CoBiMn-LDH (Fig. S1) [36, 37], proving the successful synthesis of LDH materials. High-resolution transmission electron microscopy (HR-TEM) images revealed that the size of the CoBiMn-LDH nanoparticles is 60–110 nm with the lattice spacing of ~ 0.34 nm (Fig. 2a), which corresponds to the (006) plane of the LDH crystal structure [38]. From the atomic force microscopy (AFM) image, the thickness of CoBiMn-LDH nanoparticles is found to be 6–7 nm (Fig. 2b and S2a). Energy-dispersive X-ray (EDX) elemental mapping indicated a uniform distribution of Co, Bi and Mn in the nanoparticles (Fig. 2c). After acid etching, the size of the obtained a-CoBiMn-LDH nanoparticles is slightly decreased and no obvious lattice was observed by HR-TEM (Fig. 2d), indicating its amorphous structure. As revealed by the AFM image, there was no notable change in the thickness of the a-CoBiMn-LDH nanoparticles after acid treatment (Fig. 2e and S2b). In addition, compared with CoBiMn-LDH, no Bragg reflections are observed in the XRD pattern of a-CoBiMn-LDH (Fig. 2f), further demonstrating that the a-CoBiMn-LDH nanoparticles are transformed from crystalline to amorphous [39].

(a) HR-TEM, (b) AFM and (c) EDX mapping images of CoBiMn-LDH nanoparticles. (d) HR-TEM and (e) AFM images of a-CoBiMn-LDH nanoparticles. (f) XRD patterns of CoBiMn-LDH and a-CoBiMn-LDH nanoparticles. (g) Mn 2p and (g) O 1s XPS spectra of CoBiMn-LDH and a-CoBiMn-LDH nanoparticles. (i) ESR spectra of CoBiMn-LDH and a-CoBiMn-LDH nanoparticles
To investigate the structural differences before and after acid etching, X-ray photoelectron spectroscopy (XPS) analysis was carried out (Fig. S3a and S3b). Co 2p XPS spectra showed the characteristic peaks of Co2+ 2p3/2, Co2+ 2p1/2, Co3+ 2p3/2, Co3+ 2p1/2 at 782.51, 798.42, 781.11, and 796.60 eV in both the CoBiMn-LDH and a-CoBiMn-LDH nanoparticles (Fig. S3c). Interestingly, the Co3+/Co2+ ratio of a-CoBiMn-LDH (1.35) is higher than that of CoBiMn-LDH (0.91). In the Bi 4f spectra, the peaks at 159.04 and 164.37 eV in CoBiMn-LDH could be assigned to Bi3+ 4f7/2 and Bi3+ 4f5/2 (Fig. S3d). These slightly shift to higher binding energy at 159.75 and 165.02 eV in a-CoBiMn-LDH, but in both cases the presence of Bi3+ is confirmed. The Mn 2p spectra of CoBiMn-LDH and a-CoBiMn-LDH contain binding energy peaks at 644.68, 643.37 and 641.90 eV (Fig. 2g), which were assigned to Mn4+ 2p3/2, Mn3+ 2p3/2 and Mn2+ 2p3/2, respectively, indicating that Mn4+, Mn3+ and Mn2+ existed simultaneously in the nanoparticles. O 1s spectra (Fig. 2h) reveal that different oxygen species including lattice oxygen (531.46/531.43 eV) and adsorbed oxygen (532.76/533.34 eV) existed in both CoBiMn-LDH and a-CoBiMn-LDH. It is worth pointing out that a peak from oxygen vacancies (OVs, 532.18 eV) existed in a-CoBiMn-LDH, proving the generation of abundant OVs after acid etching [40]. The existence of defects was further investigated by electron spin resonance (ESR) spectroscopy (Fig. 2i). The a-CoBiMn-LDH nanoparticles exhibit an obvious peak at G = 2.1, which is absent in CoBiMn-LDH, confirming the generation of rich defects by the etching process [41].
SDT performance
The ability of CoBiMn-LDH and a-CoBiMn-LDH in ROS generation was investigated using singlet oxygen sensor green (SOSG) [42]. After US irradiation for 6 min, the fluorescence intensity of SOSG in the a-CoBiMn-LDH group was significantly stronger (~ 3.3 times) than that of CoBiMn-LDH group (Fig. 3a and S4), indicating that the ROS generation activity could be enhanced by acid etching. The SDT properties of a-CoBiMn-LDH nanoparticles with different Bi content (10%, 20%, 30%) were also explored. It was found that the a-CoBiMn-LDH nanoparticles with 20% Bi content exhibited the strongest ROS generation performance under US irradiation (Fig. S5). In addition, the SDT properties of a-CoBiMn-LDH (if there is no special indication, a-CoBiMn-LDH refers to a-CoBiMn-LDH (20%)) under different pH environments (pH = 5.4, 6.5 and 7.4) were evaluated. As shown in Fig. S6, a-CoBiMn-LDH nanoparticles dispersed in a pH = 6.5 buffer solution (simulated tumor microenvironment) displayed the strongest fluorescence intensity, implying the system should have potent ROS generation performance in the TME. Moreover, we compared the SDT performance of a-CoBiMn-LDH with a commercial TiO2 sonosensitizer. In Fig. S4 and 3a, it can be seen that the ROS generation activity of a-CoBiMn-LDH is ~ 8.2 times that of the commercial TiO2. 1,3-Diphenylisobenzofuran (DPBF) and ESR assays further verified the superior SDT performance of a-CoBiMn-LDH than CoBiMn-LDH and TiO2 (Fig. 3b and c and S7), as the more prominent decrease in absorbance of DPBF and the stronger characteristic signal of 1O2 (1:1:1) were found in a-CoBiMn-LDH group.

(a) Fluorescence intensity of SOSG in the presence of TiO2, CoBiMn-LDH and a-CoBiMn-LDH nanoparticles under US irradiation (40 kHz, 3 W cm− 2, 6 min). (b) Normalized attenuation curves of DPBF in the presence of TiO2, CoBiMn-LDH, a-CoBiMn-LDH and a-CoBiMn-LDH + H2O2 under US irradiation (40 kHz, 3 W cm− 2, 6 min). (c) ESR spectra of 1O2 produced under different conditions. (d) Oxygen production curves of a-CoBiMn-LDH + H2O2 at pH = 7.4 and 6.5. (e) GSH consumption at different concentrations of a-CoBiMn-LDH (0, 25, 50, 75, 100 µg mL− 1). (f) Band gaps of CoBiMn-LDH and a-CoBiMn-LDH nanoparticles, and (f) their Mott-Schottky diagrams. (h) Electrochemical impedance spectra and (i) PL spectra of CoBiMn-LDH and a-CoBiMn-LDH nanoparticles
It has been reported that Mn4+ can catalyze the reaction of H2O2 to generate O2 [43]. On this basis, the catalytic activity of a-CoBiMn-LDH towards O2 generation was investigated by dissolving oxygen equipment. In Fig. 3d, the O2 concentration in a suspension of a-CoBiMn-LDH at pH values of 7.4 and 6.5 increased rapidly after the addition of H2O2, to 5.95 mg L− 1 and 12.68 mg L− 1, respectively. In contrast, no O2 generation was seen in the absence H2O2, proving the ability of a-CoBiMn-LDH to generate O2 by decomposing H2O2. In order to investigate the effect of O2 on ROS generation, DPBF was used as a probe to detect US-triggered 1O2 generation [44]. It was found that the absorbance of DPBF in the a-CoBiMn-LDH nanoparticles group decreased significantly under US irradiation (Fig. 3b). Interestingly, a more pronounced decline in the absorbance of DPBF was observed after the addition of H2O2 (Fig. 3b and S4), suggesting that O2 generation induced by the reaction between Mn4+ and H2O2 effectively promotes 1O2 production. The generation of 1O2 was further investigated by ESR spectroscopy with the 2,2,6,6-tetramethy l-4-piperidone (TEMP) probe. As presented in Fig. 3c, compared with other groups, the a-CoBiMn-LDH + H2O2 group showed the strongest peaks, further proving that O2 generation could promote the SDT performance of a-CoBiMn-LDH. In addition, since Mn4+ has been reported to react with reduced GSH, the GSH consumption capacity of a-CoBiMn-LDH nanoparticles was studied using 5,5’-dithiobis-2-nitrobenzoic acid (DTNB). As can be seen from Fig. 3e, the GSH content gradually decreased with an increase of a-CoBiMn-LDH concentration, suggesting that the nanoparticles possess GSH depletion ability. This should be conductive to reducing the clearance of ROS by GSH and promoting SDT performance.
Mechanism of ROS generation
To reveal the ROS generation mechanism, ultraviolet–visible–near-infrared (UV-vis-NIR) diffuse reflection spectroscopy was conducted to analyze the band structures of CoBiMn-LDH and a-CoBiMn-LDH. As presented in Fig. 3f and S8, the band gaps (Eg) of CoBiMn-LDH and a-CoBiMn-LDH nanoparticles were calculated to be 2.92 eV and 1.48 eV respectively, indicating a decrease in the Eg after acid treatment. The conduction band (CB) positions were determined using Mott–Schottky plots (Fig. 3g), and the CB potentials of CoBiMn-LDH and a-CoBiMn-LDH nanoparticles were measured to be − 0.80 and − 1.73 eV. Accordingly, their valence band (VB) potentials were determined to be 2.12 and − 0.25 eV. The lower CB and VB values of a-CoBiMn-LDH nanoparticles were beneficial for the excitation of e− and h+ and significantly improved their separation, with e− and h+ occupying CB and VB respectively [45]. The energy level diagram and e− transfer processes for CoBiMn-LDH and a-CoBiMn-LDH are shown in Fig. S9. Under US irradiation, the excited e− first reacts with O2 to form the intermediate ·O2−, which further combines with h+ to produce the final 1O2 [46]. Dihydrorhodamine 123 (DHR 123) that can be oxidized by ·O2− to emit a fluorescence signal at 526 nm was used as a specific probe to detect the intermediate ·O2−. In Fig. S10, the fluorescence intensity of DHR 123 was weak after the addition of CoBiMn-LDH nanoparticles, while the fluorescence intensity in the a-CoBiMn-LDH group was significantly enhanced, indicating the superior capacity of a-CoBiMn-LDH to generate ·O2−.
Electrochemical impedance spectroscopy (EIS) was utilized to investigate the e−-h+ separation ability of CoBiMn-LDH nanoparticles, where a smaller radius of the Nyquist circle means a faster electron transfer rate [47, 48]. As shown in Fig. 3h, the radius of a-CoBiMn-LDH nanoparticles in the EIS Nyquist diagram is much smaller than that of CoBiMn-LDH nanoparticles, demonstrating faster charge transfer occurred on the interface of the a-CoBiMn-LDH nanoparticles electrode. Photoluminescence spectroscopy (PL) is also an effective way to demonstrate the efficiency of e−-h+ recombination [49]. In Fig. 3i, the CoBiMn-LDH nanoparticles exhibit a strong emission peak at 371 nm. However, the peak intensity of a-CoBiMn-LDH was much weaker than that of CoBiMn-LDH, meaning that the radiative recombination of e− and h+ in a-CoBiMn-LDH was significantly inhibited, beneficial for the promotion of ROS generation.
Surface modification
The aforementioned results demonstrate the potent activity of a-CoBiMn-LDH as a sonosensitizer. Next, PEG modification was performed on a-CoBiMn-LDH nanoparticles to improve its biocompatibility [50]. In Fourier transform infrared spectroscopy (FT-IR), the characteristic peak of LDH at 1380 cm− 1 (N-O vibration of nitrate) and the characteristic bands of PEG at 950 cm− 1 (C–O–C vibration) and 846 cm− 1 (− CH2 − in-plane rocking) were found in a-CoBiMn-LDH-PEG (Fig. S11), indicating successful PEGylation. Zeta potential analysis was also conducted to verify this result. As presented in Fig. S12, the zeta potential of a-CoBiMn-LDH decreased from 13.8 ± 1.2 mV to − 10.7 ± 1.1 mV after acid etching. After PEGylation, the zeta potential of a-CoBiMn-LDH-PEG in water was − 13.5 ± 1.0 mV, which is similar to that in PBS (− 14.6 ± 1.3 mV) and high-glucose Dulbecco’s modified Eagles medium (DMEM) (− 14.0 ± 1.1 mV). The interaction between a-CoBiMn-LDH and PEG could be attributed to the Van der Waals’ force and hydrogen bonding [39]. Dynamic light scattering (DLS) analysis showed that the hydrodynamic size of a-CoBiMn-LDH-PEG is 105.1 ± 2.2 nm, larger than that of CoBiMn-LDH (90.5 ± 2.6 nm) and a-CoBiMn-LDH (78.8 ± 1.8 nm) (Fig. S13a). There was no observable size variation of a-CoBiMn-LDH-PEG after suspension in water, PBS or DMEM for one week (Fig. S13b), indicating good stability.
Evaluation of in vitro therapeutic effect on 4T1 cells.
The SDT-mediated therapeutic performance of a-CoBiMn-LDH-PEG was next evaluated in vitro. Firstly, the cellular uptake of a-CoBiMn-LDH-PEG nanoparticles by 4T1 cells was studied [51]. In Fig. S14, a strong green fluorescence signal from FITC-labeled a-CoBiMn-LDH-PEG nanoparticles was seen in cells over 24 h of incubation. Thus, a-CoBiMn-LDH-PEG nanoparticles could be effectively internalized by cells. The biocompatibility of a-CoBiMn-LDH-PEG nanoparticles on 4T1, Hela and HepG2 cells was measured using standard methyl thiazolyl tetrazolium (MTT) assays. The cytotoxicity of a-CoBiMn-LDH-PEG nanoparticles to cells was found to be negligible even at a high concentration of 200 µg mL− 1 (Fig. 4a), suggesting satisfactory biocompatibility. The biosafety of a-CoBiMn-LDH-PEG nanoparticles was further evaluated with a hemolysis assay [52]. In Fig. 4b and c, it can be seen that after incubation with various concentrations (12.5 to 200 µg mL− 1) of a-CoBiMn-LDH-PEG nanoparticles, the hemolysis rate was always lower than 5% (standard values), and thus the system does not cause significant hemolysis.
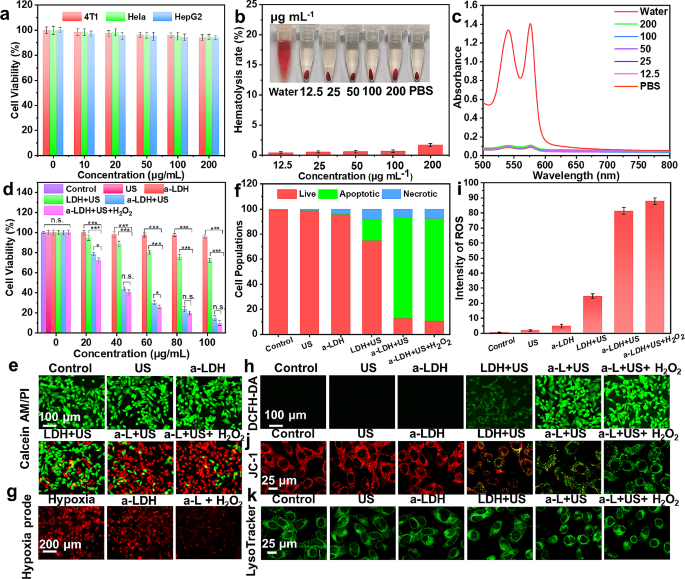
(a) Cell viability of 4T1, Hela and HepG2 cells cultured with a-CoBiMn-LDH-PEG at different concentrations. (b) Hemolysis rate of red blood cells after incubation with different concentrations of a-CoBiMn-LDH-PEG nanoparticles (12.5, 25, 50, 100, 200 µg mL− 1). Inset: representative photographs. (c) Absorbance of red blood cell supernatant treated with water, PBS and a-CoBiMn-LDH-PEG nanoparticles at different concentrations. (d) Cell viability of 4T1 cells under different conditions: (1) control, (2) US (40 kHz, 3 W cm− 2, 6 min), (3) a-CoBiMn-LDH-PEG, (4) CoBiMn-LDH-PEG + US, (5) a-CoBiMn-LDH-PEG + US, (6) a-CoBiMn-LDH-PEG + US + H2O2, and (e) corresponding Calcein-AM/PI staining images. (f) Quantitative apoptosis analysis of 4T1 cells after Annexin V-FITC/PI co-staining. (g) [Ru(dpp)3]Cl2 staining images under hypoxic conditions. (h) DCFH-DA staining images and (i) corresponding ROS quantitative analysis. (j) JC-1 and (k) LysoTracker Green staining images of 4T1 cells after different treatments. Data are expressed as mean ± S.D (n = 3). *p < 0.05, **p < 0.01, ***p < 0.001
Given the apparently biosafety of the formulations, the therapeutic effect of a-CoBiMn-LDH-PEG was investigated with 4T1 cells (Fig. 4d). The control, US alone and a-CoBiMn-LDH-PEG alone groups led to negligible cytotoxicity, while the cell viability of CoBiMn-LDH-PEG + US and a-CoBiMn-LDH-PEG + US groups significantly decreased to 72.3% and 14.8% at a concentration of 100 µg mL− 1, respectively. Moreover, the addition of H2O2 further enhanced the cytotoxic effect of a-CoBiMn-LDH-PEG under US irradiation, resulting in a cell viability of 9.8% and demonstrating the strong killing effect on 4T1 cells of a-CoBiMn-LDH-PEG in the presence of H2O2. Live (calcein acetoxymethyl ester, Calcein-AM)/dead (propidium iodide, PI) double staining analysis also evidenced the efficiency of a-CoBiMn-LDH-PEG nanoparticles for in vitro SDT. The results in Fig. 4e and S15 reveal no red fluorescence of PI (denoting dead cells) in the control, US and a-CoBiMn-LDH-PEG groups, while obvious red fluorescence was present in the CoBiMn-LDH-PEG + US and a-CoBiMn-LDH-PEG + US groups. Moreover, the brightest red fluorescence was observed in the a-CoBiMn-LDH-PEG + US + H2O2 group, consistent with the highest ROS generation efficiency being with the a-CoBiMn-LDH-PEG nanoparticles in the presence of H2O2 under US irradiation. Flow cytometry further confirmed that a-CoBiMn-LDH-PEG + US and a-CoBiMn-LDH-PEG + US + H2O2 groups successfully induced SDT-related apoptosis (Fig. 4f and S16).
Since a-CoBiMn-LDH possessed catalytic activity towards O2 generation, the hypoxia level of 4T1 cells was monitored using a red hypoxia staining reagent ([Ru(dpp)3]Cl2) [53]. Strong fluorescence signals were observed in the blank and a-CoBiMn-LDH-PEG groups, while the fluorescence intensity in the a-CoBiMn-LDH-PEG + H2O2 group decreased significantly (Fig. 4g), demonstrating that a-CoBiMn-LDH-PEG nanoparticles could alleviate hypoxia in the presence of H2O2, due to O2 generation. Furthermore, intracellular ROS levels were detected with the 2’,7’-dichlorofluorescein diacetate (DCFH-DA) probe. In Fig. 4h, no 2’,7’-dichlorofluorescein (DCF) fluorescence was observed in cells treated with DMEM, a-CoBiMn-LDH-PEG nanoparticles or US irradiation alone. However, under US irradiation, green fluorescence enhancement was induced by CoBiMn-LDH-PEG and a-CoBiMn-LDH-PEG, and the fluorescence intensity was enhanced when H2O2 was also present. ROS quantitative analysis further verified the above results (Fig. 4i).
Subsequently, the mitochondrial dysfunction of different groups was assessed using 5,5’,6,6’-tetrachloro-1,1’-3,3’-tetraethyl-benzimidazolylcarbocyanine iodide (JC-1), a mitochondrial membrane potential dye (see Fig. 4j and S17). Negligible green fluorescence from JC-1 monomers was observed in the control, US alone and a-CoBiMn-LDH-PEG groups, while increased green fluorescence was visible in the CoBiMn-LDH-PEG + US and a-CoBiMn-LDH-PEG + US groups. This the green fluorescence was significantly enhanced after the addition of H2O2, indicating extensive mitochondrial damage. We also examined the effects of different treatments on lysosomes, using the LysoTracker Green probe (Fig. 4k) [54]. The cells in the control, US alone, and a-CoBiMn-LDH-PEG groups showed green stain spots due to the lysosome wrapped in the cytoplasm, while the cells treated with CoBiMn-LDH-PEG + US showed blurred green stain spots. In the a-CoBiMn-LDH-PEG + US and a-CoBiMn-LDH-PEG + US + H2O2 groups, the green spots almost disappeared, indicating serious lysosome damage.
Biological mechanism analysis
Inspired by the above exciting results, we conducted RNA expression sequencing (RNAseq) analysis to explore the biological mechanism of a-CoBiMn-LDH-PEG killing 4T1 cells. 4T1 cells treated with a-CoBiMn-LDH-PEG + H2O2 + US and PBS were labeled as experiment and control groups, respectively. Principal component analysis (PCA) and heat maps showed significant differences in transcriptomes between the control group and the experiment group (Fig. S18 and 5a). The volcano plot results showed that a total of 4902 genes were significantly differentially expressed, of which 2452 genes were up-regulated and 2450 genes were down-regulated (Fig. 5b). In view of this, gene ontology (GO) analysis was performed to reveal the therapeutic effects of a-CoBiMn-LDH-PEG on 4T1 cells. It was found that a-CoBiMn-LDH-PEG + H2O2 + US had a significant impact on the metabolic-related functions, cellular components, and biological processes of 4T1 cells (Fig. 5c). We also conducted Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment analysis (Fig. 5d), which revealed significant changes in pathways related to oxidative phosphorylation, p53, TNF, apoptosis, and hypoxia after treatment with a-CoBiMn-LDH-PEG + H2O2 + US. Based on these results, we conducted heat map analysis of the corresponding signaling pathways (Fig. 5e), and the results showed that the treatment of a-CoBiMn-LDH-PEG + H2O2 + US significantly induced the up-regulation of apoptosis-related genes (such as Tnf, Gadd45b, and Gadd45g), and activated the p53 signaling pathway through ROS-mediated DNA damage, synergistically promoting cell apoptosis. In addition to the significant differential expression of p53 and apoptosis-related genes, the oxidative phosphorylation-related genes (Ndufa3, Cox7a2, Atp5j2) were down-regulated, indicating that a-CoBiMn-LDH-PEG + H2O2 could promote ROS production and inhibit oxidative phosphorylation in an ROS-dependent manner under US-assisted SDT treatment. Moreover, TNF signaling pathway-related genes such as Fos, Junb, and Cxcl2 were up-regulated, which increased the permeability of intracellular mitochondria and promotes the production of ROS. Furthermore, heat map analysis of HIF-1-related genes (Nos2 and Eno2) were down-regulated (Fig. S19), suggesting that a-CoBiMn-LDH-PEG + H2O2 under US irradiation could alleviate hypoxia. In addition, gene set enrichment analysis (GSEA) revealed significant positive enrichment scores for glucose catabolism, glycolysis, and apoptosis regulation (Fig. S20), demonstrating that a-CoBiMn-LDH-PEG + H2O2 + US could induce energy crisis and promote cell apoptosis.

The biological mechanism mediated by a-CoBiMn-LDH-PEG. (a) Heat map of differentially expressed genes between the a-CoBiMn-LDH-PEG + H2O2 + US (experiment) group and control group. (b) Volcano plot analysis, (c) GO and (d) KEGG enrichment analysis. (e) Heat map of expressed genes related to p53, oxidative phosphorylation, TNF, and apoptosis signaling pathways. (f) qPCR detection of gene expression (Tnf, Gadd45b, Atp5j2, and Nos2). (g) Western blot of the expression of proteins (Tnf, Gadd45b, Atp5j2, and Nos2) after different treatments and (h) corresponding quantitative analysis. Error bar represents ± S.D. (n = 3). **p < 0.01, ***p < 0.001
To validate the differential expression of RNAseq genes, real-time fluorescence quantitative polymerase chain reaction (PCR) analysis was performed. As shown in Fig. 5f, apoptotic genes (Tnf, Gadd45b) were up-regulated, while oxidative phosphorylation gene (Atp5j2) and hypoxia-related gene (Nos2) were down-regulated, validating the reliability of the above RNAseq results. Western blotting analysis was further conducted to confirm the above results. In Fig. 5g and h, compared with PBS, US, and CoBiMn-LDH-PEG + US groups, the expression of apoptotic proteins (Tnf and Gadd45b) was higher in a-CoBiMn-LDH-PEG + US group, which was the highest in a-CoBiMn-LDH-PEG + H2O2 + US group. The expression of Atp5j2 and Nos2 proteins were significantly down-regulated after a-CoBiMn-LDH-PEG + H2O2 + US treatment, which was consistent with the results of PCR analysis. Taken together, these results indicated that a-CoBiMn-LDH-PEG + H2O2 + US played an important role in promoting cancer cell apoptosis and alleviating hypoxic microenvironment.
In vivo SDT treatment
Motivated by the promising in vitro results, the therapeutic effect of a-CoBiMn-LDH-PEG was evaluated in vivo. The pharmacokinetics were first investigated. The blood circulation curve of the a-CoBiMn-LDH-PEG nanoparticles was fitted with quadratic exponents, and the calculated circulatory half-lives are 0.19 (t1/2(α)) and 8.74 h (t1/2(β)), respectively (Fig. 6a). Such a long blood circulation is conducive to the accumulation of a-CoBiMn-LDH-PEG at the tumor site. Subsequently, the biodistribution of a-CoBiMn-LDH-PEG was also studied. Hearts, livers, spleens, lungs, kidneys, and tumors were gathered at 2, 4, 8, 12, 24, and 48 h post-injection for inductively coupled plasma-atomic emission spectroscopy (ICP-AES) analysis. It was found that a-CoBiMn-LDH-PEG nanoparticles tended to accumulate in liver, spleen and tumor tissue, with the highest accumulation occurring at 8 h post-injection (Fig. 6b).

(a) Blood circulation time of a-CoBiMn-LDH-PEG in 4T1 tumor-bearing mice, quantified by determining the content of Co at each time point after injection. (b) Quantitative analysis of the biodistribution of a-CoBiMn-LDH-PEG in mice, also determined by measuring the Co concentration at various time points after injection. Error bar represents ± S.D. (n = 3). (c) Representative photos of 4T1 tumor-bearing mice given different treatments. (d) Tumor growth curves of 4T1 tumor-bearing mice after various treatments (PBS, US (40 kHz, 3 W cm− 2, 6 min), a-CoBiMn-LDH-PEG, CoBiMn-LDH-PEG + US, a-CoBiMn-LDH-PEG + US). Error bar represents ± S.D. (n = 6). (e) Representative photos of tumors on the 16th day after different treatments. (f) Average tumor weight of each group of mice after 16 days of treatment. Error bar represents ± S.D. (n = 6). (g) HIF-1α staining images of tumor sections in each group after 16 days. (h) In vivo US imaging. (i) DHE and (j) H&E, Ki-67, TUNEL, and C-Caspase3 staining analysis of tumor sections after 16 days of treatment. Each experiment was repeated three times. (k) Liver and kidney function indicators and blood cell count of mice on the 1st and 16th days after injection of PBS (control) and a-CoBiMn-LDH-PEG. Error bar represents ± S.D. (n = 3). *p < 0.05, **p < 0.01, ***p < 0.001
Subsequently, mice bearing 4T1 tumors were randomly divided into five groups (n = 6): (1) PBS, (2) US, (3) a-CoBiMn-LDH-PEG, (4) CoBiMn-LDH-PEG + US, (5) a-CoBiMn-LDH-PEG + US. Tumor size and bodyweight of the mice were recorded every two days for 16 days. Photographs of the mice at different time points and tumor growth measurements showed the significant tumor growth in the PBS, US irradiation and a-CoBiMn-LDH-PEG groups (Fig. 6c and d), whereas the tumor growth was slightly inhibited in mice injected with CoBiMn-LDH-PEG + US. In contrast, mice injected with a-CoBiMn-LDH-PEG showed complete regression of the tumor under US irradiation. The tumor tissues were gathered and weighed after 16 days of treatment. Digital images of the tumors (Fig. 6e) and the average tumor weight values (Fig. 6f) verified the significant inhibitory effect of a-CoBiMn-LDH-PEG plus US irradiation on tumor growth.
The O2-generating activity of a-CoBiMn-LDH-PEG nanoparticles was evaluated in vivo. Hypoxia-inducible factor-1α (HIF-1α) was used as a hypoxia-related marker to detect the O2 level in the tumor tissue. In Fig. 6g, obvious red fluorescence was observed in tumor tissues from the PBS- and US-treated mice, indicating the hypoxic state of the tumor. However, the red fluorescence in tumor tissues of mice injected with a-CoBiMn-LDH-PEG nanoparticles was markedly reduced, indicating that a-CoBiMn-LDH-PEG could relieve hypoxia levels by decomposing H2O2 to generate O2. In view of this, we further investigated the ability of a-CoBiMn-LDH-PEG nanoparticles for US imaging in vitro and in vivo. As shown in Fig. S21, a larger number of oxygen bubbles (white spots) were observed in the a-CoBiMn-LDH-PEG + H2O2 group compared with a-CoBiMn-LDH-PEG alone, especially at pH = 6.5, indicating the potential of a-CoBiMn-LDH-PEG for US imaging by generating O2. 4T1 tumor-bearing mice were thus injected with a-CoBiMn-LDH-PEG to conduct US imaging at different time points (see Fig. 6h). Compared with the situation before injection, the US signal contrast at the tumor site was significantly enhanced 30 min post-injection, proving that a-CoBiMn-LDH-PEG nanoparticles could not only effectively overcome tumor hypoxia, but also serve as a US imaging contrast agent [55,56,57].
Dihydroethidium (DHE) staining was performed on tumor tissues to detect ROS generation. As shown in Fig. 6i, the DHE fluorescence of the a-CoBiMn-LDH-PEG + US group was the strongest, suggesting the generation of a large amount of ROS and effective SDT performance. The in vivo therapeutic mechanism was further investigated through histological staining. Hematoxylin and eosin (H&E) staining images revealed that no significant damage was found in tumor tissues from the PBS, US and a-CoBiMn-LDH-PEG groups, while CoBiMn-LDH-PEG + US led to moderate cell apoptosis. In contrast, a-CoBiMn-LDH-PEG + US induced the most significant tumor cell apoptosis (Fig. 6j), demonstrating the most potent SDT effect of all the treatments. Ki-67 staining images (Fig. 6j) showed that a-CoBiMn-LDH-PEG markedly reduced the number of Ki-67 positive cells in tumor tissues under US irradiation, indicating its strong inhibitory effect on tumor proliferation. According to TUNEL and C-Caspase3 staining images (Fig. 6j), the a-CoBiMn-LDH-PEG + US group exhibited the strongest green fluorescence, confirming extensive cancer cell apoptosis.
In vivo biocompatibility of the a-CoBiMn-LDH-PEG nanoparticles was studied. As presented in Fig. S22, there was no significant change in the body weight of any group of mice during administration, suggesting the excellent biocompatibility of a-CoBiMn-LDH-PEG. To further evaluate the toxicity of a-CoBiMn-LDH-PEG, blood was collected on day 1 and day 16 after intravenous injection, and routine blood examinations and blood biochemical analysis were performed. It was found that blood parameters and liver/kidney function markers were not significantly different from those of the PBS group (Fig. 6k), indicating negligible blood toxicity of a-CoBiMn-LDH-PEG. H&E staining images of the heart, kidney, liver, lung, and spleen after the end of the treatment period showed that there was no obvious physiological abnormality or difference between the a-CoBiMn-LDH-PEG and PBS groups (Fig. S23), again indicating low toxicity. The survival time of mice in each group was also recorded. In Fig. S24, it can be seen that the a-CoBiMn-LDH-PEG + US treated mice remained healthy for 60 days after treatment, without tumor recurrence. In contrast, the other groups of mice died to various extents over this period of time, confirming the therapeutic efficiency of a-CoBiMn-LDH-PEG nanoparticles under US irradiation. The Co content in urine and feces was determined by ICP-AES to explore the metabolism of a-CoBiMn-LDH-PEG nanoparticles in vivo. As shown in the Fig. S25, a high concentration of Co was detected at 8 h post-injection and this then gradually decreased, demonstrating that a-CoBiMn-LDH-PEG nanoparticles could be metabolized effectively and excreted in the feces and urine. In summary, the above results prove that a-CoBiMn-LDH-PEG nanoparticles possess good biocompatibility, potentiating its further application as a highly active sonosensitizer for SDT.


